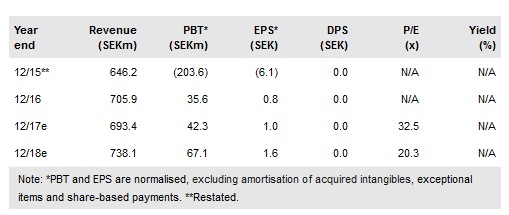
Orexo AB (ST:ORX) made significant progress in 2016 towards becoming a sustainable speciality pharma company, with continued Zubsolv revenue growth contributing to a first full year of profitability. Key achievements included the ex-US global Mundipharma licence deal for Zubsolv (EMA filing in Q4), and the first large exclusive Medicaid FFS contract in Maryland. Despite this, and ahead of major newsflow in 2017, IP infringement litigation remains a stock overhang with Orexo trading near 52-week lows. We believe the market’s view of Zubsolv’s prospects is too pessimistic. Our new assumptions, updated for evolving market dynamics, generate a SEK3.16bn or SEK91/share valuation (vs SEK4.54bn or SEK131/share).
FY16 operational review
FY16 delivered the Mundipharma deal and Europe filing (potential approval Q417), completion of the RESOLV study, approval of the lowest Zubsolv dose strength and grant of two new patents. Disproportionate share of growth with C275 physicians is being captured by Zubsolv as new legislation is implemented. The outcome of the first IP challenge precludes Actavis (NYSE:AGN) from generic launch pre-2019; Orexo has appealed the invalidity of the ’330 patent (ruling late 2017/early 2018).