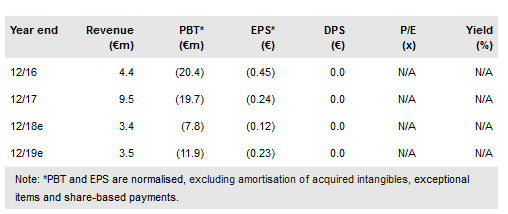
Onxeo (CO:ONXEO) lead asset, AsiDNA, is currently being tested in a Phase I DRIIV-1 trial in patients with advanced solid tumours (n=36). Interim results were announced recently, based on which Onxeo will initiate a further Phase Ib/II development programme, likely combining AsiDNA with other standard-of-care drugs that have shown the highest potential in preclinical models. The most interesting combination seems to be with PARP inhibitors. These activities are being funded by cash raised from the recent Beleodaq royalty stream monetisation ($7.5m) and equity financing agreement, both in June 2018. Our valuation is €172m or €3.3/share.
Moving to Phase Ib/II after DRIIV trial interim results
AsiDNA is part of the proprietary, novel platON platform, a major R&D expansion announced in October 2017, and is based on decoy oligonucleotides, which makes it a unique drug with no close comparators with respect to mode of action. In a broader sense, the platON platform belongs to the so-called DNA damage response (DDR) technology, a domain in which recently marketed PARP inhibitors also belong. On 5 November 2018, Onxeo reported interim results from the first clinical DRIIV-1 trial where AsiDNA was administered intravenously. After testing the first three dose levels (out of six), no serious drug-related side effects were seen and the maximum tolerated dose has not been established yet. Activity and tumour biomarker analysis allowed Onxeo to conclude that mechanism of action in humans after systemic administration has been proven and the company is now planning further Phase Ib/II studies, which will likely include combination treatments, with substantial focus on PARP inhibitors and chemotherapies.
To read the entire report Please click on the pdf File Below..