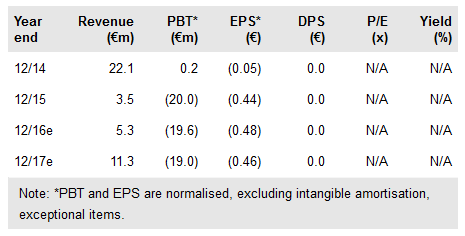
With its acquisition of DNA Therapeutics, Onxeo (PA:C4X) has added a clinical stage signal-interfering DNA (siDNA) technology, which has already been tested in a Phase I trial for melanoma and demonstrated an attractive safety profile and preliminary anti-tumour activity, when administered locally. The new product called AsiDNA is a first-in-class and differentiated asset in the up-and-coming DNA repair interference field. Our increased valuation of Onxeo is €343m.
Acquisition adds clinical stage asset
Onxeo has acquired Paris-based private biotech company DNA Therapeutics for an upfront payment of €1.7m by issuing new shares (1.4% of the total outstanding before the acquisition). An additional €1m in shares or in cash is due once lead-product AsiDNA enters a Phase II trial, which will explore AsiDNA administered systemically. Future commercial royalty payments could total up to €25m per indication. The timing of the initiation of Phase I with AsiDNA has not been announced yet, but we believe it could be around the end of 2016 or early 2017, which we use in our model.
To read the entire report Please click on the pdf File Below