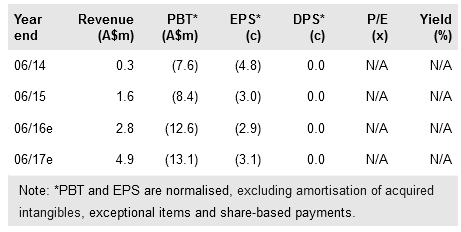
Novogen Ltd (AX:NRT) has unwound its CanTx JV with Yale and resumed 100% ownership of its lead superbenzopyran Cantrixil, as it prepares to enter the clinic in ovarian cancer patients in Q416. Separately, its anti-tropomyosin (ATM) drug Anisina, which shows strong synergy with standard-of-care anti-mitotic drugs, is scheduled to enter the clinic in H216 or H117 (pending successful completion of toxicology studies). With A$38m cash, we believe that Novogen is funded through FY18. Our valuation is virtually unchanged at A$113m (vs A$115m), with the benefit of resuming 100% ownership of Cantrixil offset by higher forecast G&A expenses.
Cantrixil on track to enter clinic in Q416
Novogen expects to enrol the first patients in Q416 in a Phase I trial of intra-abdominal Cantrixil in ovarian cancer. It has appointed Quintiles as CRO to support execution of the study at centres in Australia and the US, and plans to submit an IND to the US FDA in August. A poster presenting the recently completed Cantrixil preclinical toxicology studies at AACR in mid-April concluded that “a dose should be tolerated in humans which is slightly higher than the efficacious dose for the three times weekly regimen in a mouse disseminated ovarian cancer model”.
To read the entire report Please click on the pdf File Below