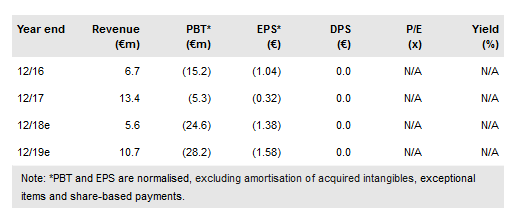
Xadago for Parkinson’s disease (PD) is now available in 14 European countries through partner, Zambon. Additionally, the drug is available in the US market (launched in H217 by sublicensee US WorldMeds). Newron Pharmaceuticals SpA (SIX:NWRN) reported revenues of €13.4m in FY17 (+100%) driven by royalty and milestone payments for Xadago and an operating loss of €4.3m (-71%). We expect pipeline progression in FY18: sarizotan (Rett’s syndrome, RS) and Evenamide (schizophrenia) highlight a diverse and innovative CNS-based R&D portfolio. We value the company at CHF758m.
Xadago ramping up slowly but steadily
Newron reported €2.9m in Xadago royalties from commercial partner Zambon in FY17 (FY16: €1.7m); the product is now available in 14 European countries. US FDA approval on 21 March triggered a €11.3m milestone payment to Newron from Zambon. FY18 sales should benefit from the ongoing European market rollout by Zambon and a full year on the US market. Newron and Zambon plan to initiate a Phase III Xadago in PD-related dyskinesia trial later this year.
To read the entire report Please click on the pdf File Below: