Nanobiotix (NANO:Euronext Paris) is a French nanotechnology company developing radiotherapy enhancers for the treatment of cancer. Lead product NBTXR3 is currently in Phase I clinical development in Europe and is partnered with PharmaEngine in Asia-Pacific.
First clinical data have shown good efficacy, with two patients at the lowest dose achieving 95% tumour necrosis, which compares favourably to historic radiotherapy data. In addition, NBTXR3 demonstrated good tumour diffusion and no leakage to surrounding healthy tissue. No serious treatment related adverse events were observed. These data increase our confidence of success in NBTXR3, raising our valuation to €10.6/share.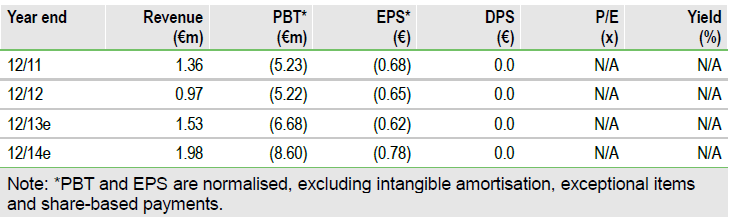
First signs of efficacy in humans
Efficacy data from the ongoing Phase I soft tissue sarcoma (STS) trial were presented around ASCO. These demonstrated nearly 95% tumour necrosis in two patients at the lowest dose (n=6), compared to historic reference data where 8% achieved this with conventional radiotherapy. The vast majority of patients also experienced tumour shrinkage. Higher doses could have an even greater benefit.
No serious adverse events even in large tumours
NBTXR3 was shown to have good tumour diffusion with no leakage to surrounding healthy tissue. No serious treatment-related adverse events were observed, including an 82 year-old with a tumour volume of nearly 3.7l injected with 184ml (5%) of NBTXR3. This is likely to be much larger than doses needed to treat patients with, for example, head and neck cancer.
Increased confidence in NBTXR3 adds €2/share
These data provide first proof-of-concept, and increase our confidence in NBTXR3. Our probability of success in Europe and in the Asia-Pacific moves to 40% (from 30%). In the US, our probability is increased to 30% (from 25%), with the lower probability reflecting the risks of successfully executing an out-licensing deal, and the additional clinical trials required before approval as a drug in this region.
Next development steps
The ongoing Phase I STS trial should move to the next 10% dose level during H213 following the upcoming data safety monitoring committee meeting in July. A Phase I head and neck trial is due to start in Europe in the near term. Partner PharmaEngine is due to start Phase I trials in the Asia-Pacific this year.
Valuation: Increased rNPV of €113m
We have increased our valuation >25% to €113m or €10.6/share based on a risk-adjusted NPV analysis. This suggests >60% upside to the current share price.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Nanobiotix: Giant Leap Forward From Initial Small Steps
Published 06/10/2013, 08:44 AM
Updated 07/09/2023, 06:31 AM
Nanobiotix: Giant Leap Forward From Initial Small Steps
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.