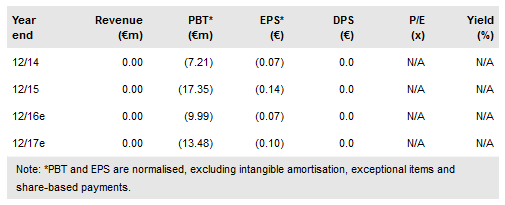
Kiadis Pharma BV (AS:KDS) has announced an update on the second dose Phase II study of ATIR101 in patients with haematological malignancies to investigate whether a second dose could be administered if needed. There were cases of grade III/IV Graft vs Host Disease (GVHD) in some of the six patients who underwent a second dose of the product. Kiadis shares initially dropped 8% on the news, but have since recovered some of the losses. We see no impact on our valuation or forecasts following this news, as the market opportunity we forecast is predicated on a single dose of ATIR101, which will be investigated in the upcoming Phase III trial. Furthermore, safety has already been established for the single dose in previous studies. This second dose trial was purely to see if a second dose strategy could improve patient outcomes. We maintain our valuation of €383.2m.
Study update: No second dose for now
Data from an exploratory Phase II study (n=15) have been announced. So far, at least one dose of ATIR101 has been administered to 10 patients and safety has been consistent with the previous Phase II study, showing no grade III/IV GVHD. A second dose of ATIR101 was administered to six patients to investigate whether this approach could further improve the outcome of haematopoietic stem cell transplantation (HSCT). Some of the six patients experienced grade III/IV GVHD, which prompted the Independent Data Monitoring Committee (IDMC) to recommend proceeding with the study with only one dose. The IDMC review was a pre-planned safety measure of the study protocol.
To read the entire report Please click on the pdf File Below