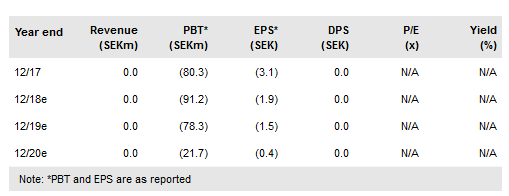
Immunicum AB (ST:IMMUN) is a Sweden-listed, clinical-stage immunoncology (IO) company that develops allogeneic dendritic cell (DC) technologies. Its first clinical product, ilixadencel, is in Phase I and II combination studies in several solid tumour indications. Two studies are expected to report in 2019; the first is the Phase II MERECA data in renal cell carcinoma (in combination with sunitinib). Interim data from the Phase I/II multi-indication study where ilixadencel is being tested in combination with a checkpoint inhibitor are also expected in H219. We initiate coverage of Immunicum with an rNPV valuation of SEK1.13bn or SEK22.1 per share.
An ‘off-the-shelf’ immune primer
Ilixadencel is an immune primer that uses activated DCs. It should not be confused with a DC therapy or DC vaccines. Immunicum’s DCs are allogeneic (from healthy donors) and, as such, induce an anti-tumor response. Injected directly into the patient’s tumor, their role is to attract and specifically activate the patients’ immune system to recognize and kill the tumour cells. Ilixadencel is being developed for advanced solid tumors and combined with drugs known to inhibit tumour-dependent immune suppression at different points in the cancer immunity cycle.
To read the entire report Please click on the pdf File Below: