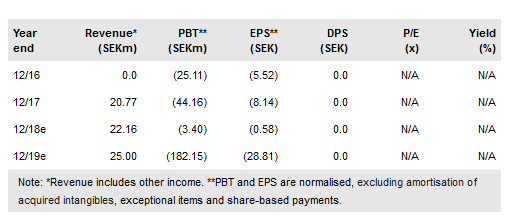
Xbrane Biopharma AB (ST:XBRANE) now focuses on developing high-margin biosimilar products. In July, Xbrane did a deal with STADA on Xlucane, its Lucentis biosimilar and gained an upfront fee of €7.5m; management estimates that Xbrane’s 50:50 profit share could be worth up to €100m per year. STADA will market Xlucane. Xbrane aims to start clinical development in Q119. In drug delivery, Spherotide bulk sales to Iran were SEK14m to June 2018. In Europe, the key market, Spherotide development requires a partner to fund trials. The Chinese deal with CR Pharma completed in February, bringing a SEK13m fee. Our valuation has been revised to SEK581m.
Xlucane the core focus with STADA partnering
Xlucane is developing a biosimilar of Lucentis (ranibizumab, Roche/Novartis; 2017 sales: $3.3bn) to treat wet age-related macular degeneration (wAMD). In July 2018, it was globally partnered with STADA, a German company with €2.3bn 2017 sales, a large generic portfolio and strong European sales capability. Development costs and post-marketing net profits are split 50:50. Competitors include bioeq (partnered with Formycon) with Phase III data and Samsung (KS:005930) Bioepis with a Phase III trial underway. Xbrane and STADA plan to be ready for a European launch in 2022 on patent expiry with the planned Phase III clinical trial being initiated in Q119.
To read the entire report Please click on the pdf File Below..