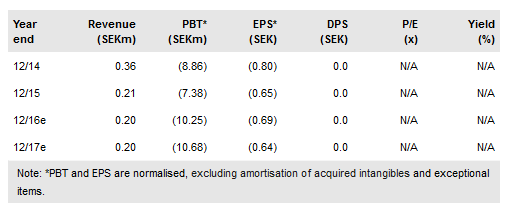
Immunovia (ST:IMMNOV) has taken key steps in both opportunities being pursued for its IMMray PanCan-d test for the early detection of pancreatic cancer. In diabetes, a collaboration with the Early Detection Research Network (EDRN), part of the US National Cancer Institute (NCI), is an important step forwards as it could lead to the start of the required clinical study. Diabetes is the larger of the two indications with 3.4 million newly diagnosed diabetes patients every year in the US and EU. In high-risk pancreatic cancer patients, Immunovia has now started a prospective clinical trial, as expected. This is a smaller opportunity (c 200k patients) but could generate initial revenues in 2018. Our valuation remains SEK155.2/share.
Immunovia delivers agreement with NCI
Immunovia has made important first steps towards its strategy of pursuing new onset diabetic patients by signing a memorandum of understanding (MoU) with the NCI EDRN. The goal of the collaboration with EDRN is to validate biomarkers to improve early diagnosis of pancreatic cancer, in particular in patients over 50 years old with new onset diabetes. These patients have eight times more risk to develop pancreatic cancer. We believe that this collaboration could lead to the start of a formal clinical trial to test IMMray PanCan-d in this population in order to gain regulatory approvals for the test. We anticipate that any such trial could involve 5,000 to 10,000 patients, analysed at least twice a year over three years. Immunovia recently raised c SEK218.6m to advance the diabetic opportunity.
To read the entire report Please click on the pdf File Below