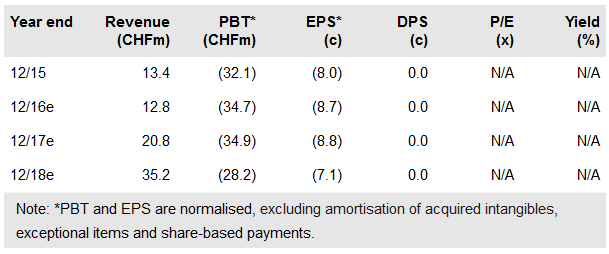
Evolva Holding SA (SIX:EVE) has received two important pieces of good news: first, on 26 May it was granted a pivotal patent for the commercial production of fermentation-derived steviol glycosides, which is key to the expansion of stevia and demonstrated that Evolva is ahead of the competition. Second, on 1 June it was issued with a US FDA GRAS no objection letter for EverSweet. This news should serve as a reminder that EverSweet remains a key part of the Evolva investment case.
Ahead of the competition
On 26 May Evolva announced it had been granted a European patent which is key to the commercial expansion of stevia sweeteners. The news allayed fears in the market that Evolva was lagging its competitors in terms of technology and the protection of its stevia intellectual property (IP), and in fact demonstrated that it is ahead of the competition. Separately, we note that on 1 June US Customs and Border Protection (CBP) issued a detention order on stevia produced by PureCircle in China based on information that forced labour is involved. This has been disputed by PureCircle, which is working to have its shipments released. While we cannot attempt to comment on whether or not forced labour is involved, this highlights the issue that the current methods of stevia production (directly from the stevia leaf) rely on complex supply chains which are hard to trace. Evolva’s fermentation method will simplify the supply chain and improve transparency, which should be of significant interest to the food and beverage manufacturers.
To read the entire report Please click on the pdf File Below