Consort Medical Plc (LON:CSRT) and Astra Zeneca’s ‘significant’ new respiratory device supply agreement highlights the ongoing strength of Bespak’s metered dose inhaler (MDI) franchise. MDIs accounted for c 51% of the Bespak division’s total product revenues of £117.2m in FY16.
This new multi-year agreement includes scale-up and supply of proprietary pMDI valves and actuators, which are critical components of AstraZeneca’s approved COPD (chronic obstructive pulmonary disease) therapy, Bevespi Aerosphere.
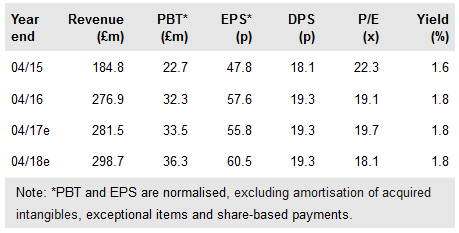
VAL100: New commercial supply agreement with AZ
Bespak will scale up and supply proprietary pressurised MDI (pMDI) valves and actuators to Astra Zeneca for assembly into the Aerosphere pMDI device. These components will be manufactured in Bespak’s existing King’s Lynn facility.
This project, termed VAL100, becomes the 15th development project in Bespak’s pipeline; of these, four are MDI focused. Terms are undisclosed although we note that according to Consort, each project represents ≥£3m peak revenue potential.
To read the entire report Please click on the pdf File Below