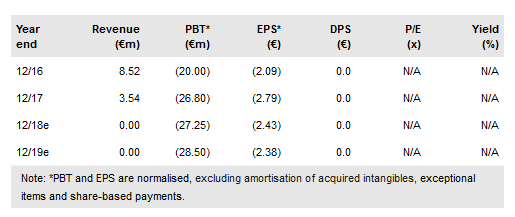
The FDA’s sign off on Celyad's (NASDAQ:CYAD) first clinical trial design for its allogeneic NRK CAR T-cell therapy (CYAD-101) is an important milestone. The study, possibly staring in Q4 2018, mirrors the current colorectal SHRINK trial a combination of autologous CYAD-01 therapy with FOLFOX chemotherapy. This gives Celyad the lead in a mass-market solid cancer where allogeneic therapy is likely to be essential. The indicative value has been increased to €1,090m (€89 per share) from €1,040m (€84 per share) pending further data.
Leading position in solid cancers and allogeneic cells
NKR CAR T-cell therapy is a generalised approach targeting “stressed” cancer cells, for example, after chemotherapy like FOLFOX. Combined with an allogenic approach, this offers a more affordable, rapid -response potential therapy for many thousands of patients. This gives Celyad the dominant position in solid cancers, an area other CAR therapies find hard to access (see report). Celyad is already evaluating autologous (customised) CYAD-01 with FOLFOX in the SHRINK trial and the approved IND mirrors that trial design. SHRINK seeks to optimise the safe CYAD-01 dose and dose schedule. We are not aware of any other allogeneic CAR T-cell therapy in solid cancers and only three in haematological cancers.
To read the entire report Please click on the pdf File Below: