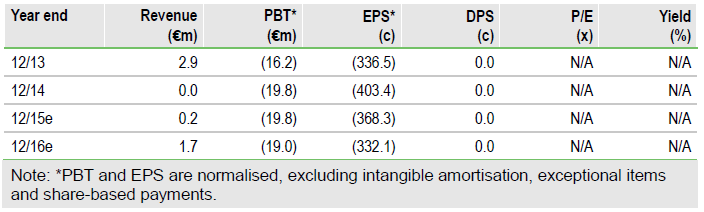
Green light to conclude feasibility study
Carmat (PA:ALCAR) has obtained regulatory authorisation to conclude the feasibility study of its artificial heart and to adopt a new clinical protocol enabling patients with less severe heart failure to participate. This should pave the way for a CE mark study in 2016 and could lead to a launch in CE mark regions in H217. Our valuation is raised from €591m to €611m (€129/share).
Regulators approve final step in feasibility study
The French health regulator (ANSM) and person protection committee (CPP) have given Carmat the go-ahead to finalise the four-person feasibility study of its novel artificial heart. This follows a review of Carmat’s proposed solutions to address the malfunctioning of the device in an earlier patient, caused by a minute leak of blood into the activating liquid of the prosthesis. Carmat aims to start a pivotal study in early 2016, which may pave the way for a CE mark grant and launch in 2017.
To Read the Entire Report Please Click on the pdf File Below