Carmat (PA:ALCAR) has received approval from the health regulatory authorities in France to start a CE mark enabling a pivotal trial for the Carmat bioprosthetic heart. We expect recruitment to start in the coming weeks, which could lead to commercialisation in Europe by H218.
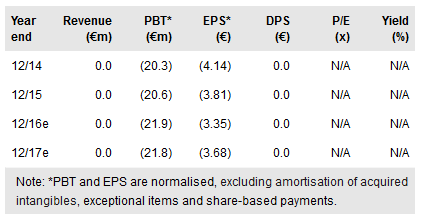
Following this clearance, we have raised our Carmat heart probability of success estimate for the EU market to 35% (from 30% previously). This raises our rNPV valuation to €747m (up from €651m), or €133.00 per share after including an estimated mid-2016 net cash position of €41.3m.
Pivotal trial expected to start in H216
In addition to patients with end-stage biventricular heart failure, patients who are eligible for a heart transplant but who have a very low likelihood of receiving a donor heart will be allowed to participate in the trial.
This inclusion criteria is more relaxed than that of the prior four-patient feasibility study (completed in early 2016), which was limited to terminal-stage biventricular heart failure patients and/or those in an immediate life-threatening situations. Carmat is also working on obtaining clearances to enrol patients in other European countries.
To read the entire report Please click on the pdf File Below