Ascelia Pharma AB (ST:ACELP) recently announced its maiden interim financial results as a public company. Q319 operational expenditure and net loss has increased versus Q318 due to the Phase III study preparation and IPO costs. It is interesting to see a company with a clinical pipeline appear on the public market. Ascelia’s first product is Phase III ready with a sophisticated regulatory strategy.
Interim financial results
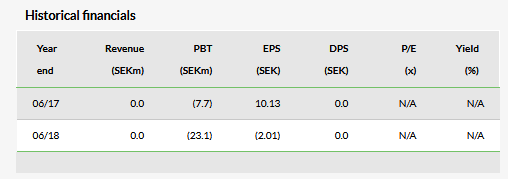
The maiden financial report of a newly listed company is largely cosmetic and an update of the recent prospectus. In Ascelia’s case, the Q319 results announcement noted an increased operating loss of SEK11.7m (SEK4.4m in Q318) due to the preparations for the Phase III study and the IPO costs. R&D and administrative costs comprised virtually all the SEK11.6m loss for Q319, excluding the R&D tax rebate. Ascelia reported a robust Q319 SEK219m cash position (SEK55m at the end of Q318) after raising SEK180m at the IPO at SEK25 per share. The Mangoral Phase III study is now fully funded with results expected around the end of 2020.
Lead product Mangoral about to start Phase III
At a point in the market where US life science IPOs are often preclinical, it is refreshing to see a European emerging specialty pharma oncology company come to the public markets with a mature clinical pipeline. The lead product – the imaging agent Mangoral – is being studied in the detection of liver tumours. Phase III for Mangoral has a primary endpoint of the detection of lesions by MRI (with and without Mangoral) and the study only has a 72-hour safety follow up. This means that Mangoral could have a label that includes both primary and secondary liver cancer detection. The Phase III study is not dependent on a long follow up (which would be costly) or a liver biopsy to confirm the cancer, which tends to restrict recruitment in drug studies in other liver indications. Outside oncology, non-alcoholic steatohepatitis is a potentially large market that is being restrained by its definitive diagnosis. If Mangoral can detect both fibrotic and cancerous liver lesions, it will be a significant advance.
Valuation: Not yet appreciated by the market
Recent IPOs may not be efficiently valued. However, with an enterprise value of only SEK244m, one Phase III-ready imaging product and one Phase II-ready oral oncology drug (Oncoral for gastric cancer) and a market capitalisation that is half cash, this appears to be a very low pipeline valuation. The average EV for three recent preclinical US IPOs is SEK1.82bn ($184m), which highlights the valuation disparity between US and European life science companies.
Business description
Ascelia Pharma is an oncology drug development company. It develops drugs to enhance the detection of liver cancer by MRI (Mangoral) and for the treatment of gastric cancer (Oncoral). With Mangoral about to start Phase III and Oncoral ready for Phase II, Ascelia is emerging as a specialty oncology company that aims to market its own products.
Bull
A clinical pipeline that includes a Phase III- and a Phase II-ready product.
As an MRI imaging-enhancing agent, Mangoral appears lower risk than a therapeutic product with a sophisticated regulatory strategy.
The recent IPO provides the financial resources for Mangoral’s development with results around the end of 2020.
Bear
As a new IPO, Ascelia is largely unknown and trading in its stock is relatively illiquid.
Ascelia’s second product Oncoral is a therapeutic oral reformulation of cancer drug irinotecan, which is a higher-risk product than Mangoral.
Pipeline progress will be associated with additional funding requirements.