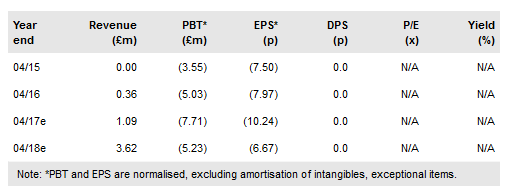
Angle's (LON:AGLE) H117 business update revealed that R&D activities are progressing well, while the company booked further research use sales. The first prospective clinical studies with Angle’s liquid biopsy system Parsortix in ovarian cancer reported positive early evaluation results and are due to report headline data in Q217. Our SOTP-based valuation is increased modestly to £140.3m or 188p/share. The main 2017 catalysts are the results from the ovarian cancer studies; an acceleration of research use sales; any new data from Angle’s multiple KOLs and customers investigating Parsortix; and progress with the FDA analytical and clinical studies.
Positive early evaluation in ovarian cancer studies
Currently two clinical studies (n=200 each) are running in parallel in Europe and the US. They aim to explore Parsortix’s efficacy in triaging women with ovarian masses before surgery to determine whether the tumour is benign or malignant, allowing for appropriate intervention. On 26 January 2017, Angle announced a positive interim evaluation of the ovarian cancer studies after the first 50 patients were enrolled in each trial. Headline results from both studies are expected in Q217. The trials are based on the work of Angle’s partners at the Medical University of Vienna, which in 2015 reported from a small trial “unprecedented sensitivity and specificity” of the use of Parsortix for ovarian cancer detection, with reported sensitivity of 80% using seven RNA markers and 100% using 30 markers, while specificity was 100% in both cases.
To read the entire report Please click on the pdf File Below