Allergy Therapeutics (AGY.L) intends to become a top-three player in the global AIT (allergy immunotherapy) market; recent and upcoming regulatory catalysts should support this, driving future revenue growth. The FDA clinical hold lift on Pollinex Quattro (PQ) Grass permits Allergy to go ahead with US plans to secure a partner within the next 12 months. PEI feedback (and potential approval) of the German PQ Grass MAA is expected in Q412, which will allow commercial marketing in Germany and the initiation of filings across Europe under the Mutual Recognition Procedure (MRP).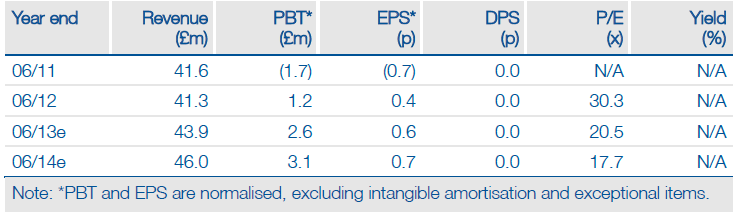
US: Aiming to close PQ partnership in 8-12 months
The formal process will start imminently with non-confidential PQ data being sent to a shortlist of 25 potential licensing partners. A smaller group, identified in 10-12 weeks, will undertake more extensive due diligence under CDA with the aim of closing a deal within 8-12 months. The partner is expected to fund pivotal trials and to provide sales infrastructure. Best case, PQ Grass could be on the market in three years.
Europe: PEI feedback to expand PQ EU penetration
Germany is the largest AIT market globally; PEI approval of PQ Grass would allow increased promotion but with an immaterial price increase. More importantly, following approval, PQ Grass registrations will be sought under the MRP in new markets where it is not available (eg Scandinavia, Central Europe). The MRP process could take a year, thus PQ could become more widely available by early-2014. Initial feedback on the 10 MAAs filed in 2011 should also start to come through from end-2012 onwards.
Financials: A third consecutive year of operating profit
Gross revenue grew 2%, but net revenue was affected by German market dynamics; ex-German growth was 9% (constant currency). Named patient PQ sales grew 2.8% to £21.4m. Lower overheads supported a third year of operating profit. Net debt of £620k reflected the April balance sheet restructuring. Guidance remains double-digit revenue growth in the next five years and pre-R&D EBITDA c 20% of sales. We forecast lower net revenue growth in FY13/FY14, but continued operating profitability.
Valuation: £55-70m ex-US DCF vs £48m EV
The US is a significant additional opportunity, which is not yet factored into the Edison revenue model. We intend to include this when a US partner is secured and there is visibility on both development timelines and deal economics. Allergy’s EV of c £48m (market cap of £48.8m minus net debt of £0.6m at end-June 2012) compares with an implied £55-70m DCF-based valuation range (12.5% discount rate; 2.5-5.0% TGR).
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Allergy Therapeutics: Third Consecutive Year Of Operating Profits
Published 10/03/2012, 03:07 AM
Updated 07/09/2023, 06:31 AM
Allergy Therapeutics: Third Consecutive Year Of Operating Profits
Potential to boost PQ sales
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.