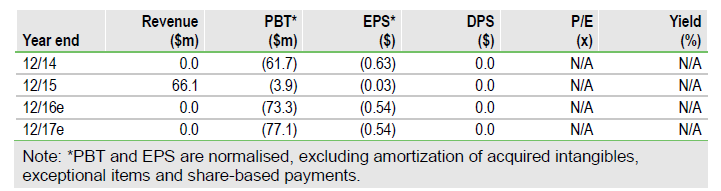
Achillion Pharmaceuticals Inc (NASDAQ:ACHN)
Triple regimen is a potential six-week treatment
Updated interim results from the Phase IIa dose ranging study ALS-022335, simeprevir and odalasvir for the treatment of genotype 1 hepatitis C virus (HCV), were announced at the European Association for the Study of the Liver conference in September 2016. The study showed a sustained virologic response at 12 weeks post treatment (SVR12) or longer of 100% for all patients (n=60) who received three drugs. Moreover, one cohort (n=20) showed 100% SVR12 following only six weeks of treatment.
Triple regimen works in 100% of those tested
A total of 80 patients were treated across four arms of the open-label study. The only arm in which patients did not achieve 100% SVR12 was one omitting simeprevir. SVR12 has been used as an approval endpoint (in addition to durability of response) for all the other recently approved HCV treatments.
To read the entire report Please click on the pdf File Below