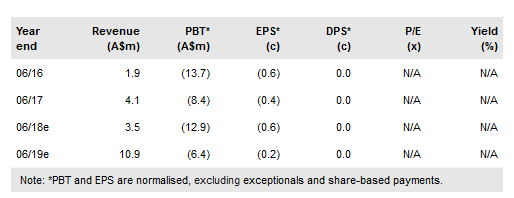
Immutep Ltd ADR (NASDAQ:IMMP) has entered a clinical trial collaboration and supply agreement with Merk & Co (MSD) for a Phase II study to evaluate eftilagimod alpha (IMP321) plus Keytruda in lung, head and neck and ovarian cancers. It is positive to see Immutep collaborating with a leading immunotherapy company, with three new indications added to its ongoing studies in breast cancer and melanoma. It has raised A$6.9m through a placement and has opened a share purchase plan to raise up to A$10m. We increase our valuation to A$439m (vs A$272m) or A$0.14/share (vs A$0.12/share).
Phase II in collaboration with Merck (NYSE:MRK) to begin H218
Immutep and Merck will collaborate in the TACTI-002 Phase II study of IMP321 plus Keytruda in up to 120 patients with non-small cell lung cancer (NSCLC), head and neck cancer or ovarian cancer. In clinical studies IMP321 has been shown to stimulate the immune system and turn “cold” tumours “hot”. There is good potential for response rates in these three cancers to be increased by combining the immune activation activity of IMP321 with an immune checkpoint inhibitor such as Keytruda, particularly in patients with low levels of PDL1 expression in tumours.
To read the entire report Please click on the pdf File Below: