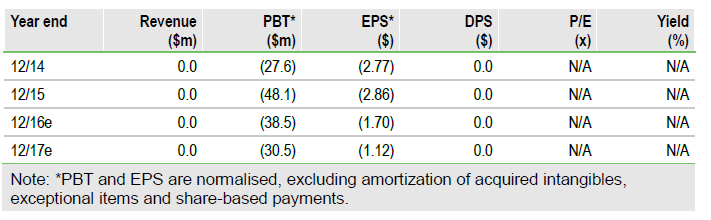
Fibromyalgia setback, focus on PTSD
Tonix Pharm (NASDAQ:TNXP) reported that the Phase III trial of TNX-102 SL for the treatment of fibromyalgia did not reach its primary endpoint of the proportion of 30% responders compared to placebo (p=0.095). The company also announced that it will discontinue the fibromyalgia program and focus its resources on advancing TNX-102 SL for post-traumatic stress disorder (PTSD), about to enter Phase III in Q117.
Fibromyalgia results skewed by discontinuations
The active arm of the trial showed a significant increase in the number of discontinuations (22.5% vs 13.6% for placebo), many of which (5.7% vs. 1.2%) were due to “withdrawal of consent” which could be for any reason including patients moving away. These discontinuations counted as non-responders as dictated by the statistical plan. The results would have been significant if these discontinuations were not counted as failures (p=0.012).
To read the entire report Please click on the pdf File Below