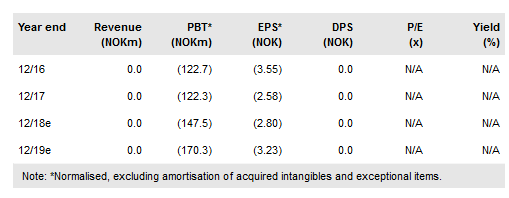
Q417 saw Targovax (LON:0RISol) release of several reports indicating a good safety profile and consistent immune activation signs by ONCOS-102, an oncolytic virus for melanoma and mesothelioma, and TG02, a neo-antigen vaccine for colorectal cancer. 2018 catalysts include full two-year survival data from the pancreatic cancer trial (Phase I/II) with TG01 and interim data readouts from the melanoma (Phase I) and mesothelioma (Phase Ib/II) trials with ONCOS-102. Our valuation is marginally higher at NOK1.78bn or NOK33.8/share.
Early consistent immune activation signs
Targovax runs four trials across its two platforms and supports an additional two trials led by partners. Targovax’s most advanced Phase I/IIa trial with TG01 for pancreatic cancer is due to report final data and the company has presented potential design of the next trial, which will be part of the registration programme. According to early findings, both ONCOS-102 and TG02 have been found to consistently activate the innate and adaptive immune system. While the patient numbers were small at the time of the analysis, Targovax expects to report more mature interim data with ONCOS-102 in both indications in 2018 (Exhibit 1).
To read the entire report Please click on the pdf File Below: