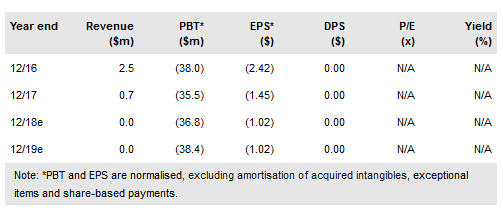
On the YE FY17 conference call, Sunesis Pharmaceuticals Inc (NASDAQ:SNSS) provided revised guidance on the dosing portion of its ongoing Phase Ib/II study of vecabrutinib (SNS-062) in chronic lymphocytic leukemia (CLL) and other B-cell cancers. The final dose is expected to be reached in fall 2018 (revised from mid-2018) due to an on-protocol expansion of the second (50mg) dosing cohort because of a dose-limiting adverse event (AE). At this time we do not consider this delay or the AE to be material to the success of the program.
Cohort expansion triggered for 50mg dose
The dose escalation is of a traditional 3+3 design, in which an additional three patients are enrolled if a dose-limiting toxicity (DLT) is observed in the initial three person cohort. A DLT is defined as an AE that prevents the patient from continuing dosing, although this may be an isolated event. The 3+3 design is structured to adapt to such unforeseen events to find the true maximum tolerated dose, although it does trigger a delay before higher doses can be assessed.
To read the entire report Please click on the pdf File Below: