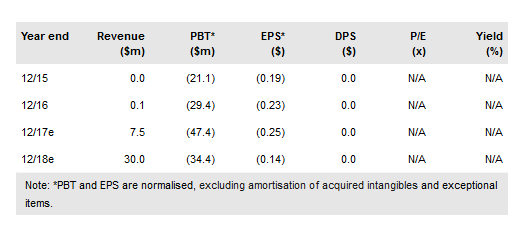
In October 2017, Redhill Biopharma Ltd (NASDAQ:RDHL) announced that one of its core assets, BEKINDA, significantly alleviated symptoms of patients with diarrhoea-predominant irritable bowel syndrome (IBS-D) in a Phase II trial. Although not comparable directly, the data look good in relation to two other recent drugs, Viberzi and Xifaxan, which had combined sales of $382m in 2016 after the launch in 2015 for this indication. We have increased our success probability for BEKINDA in IBS-D and now value RedHill at $449m or $21.1/ADS.
BEKINDA: Primary endpoint met in Phase II trial
BEKINDA (formerly RHB-102) is a once-daily, oral formulation of ondansetron. The Phase II trial was a randomised, two-arm study that enrolled 126 patients split 60:40 to receive either BEKINDA 12mg or a placebo for eight weeks. The primary endpoint was stool consistency compared to the baseline, as per the FDA guidance definition. 54.7% of patients in the active arm responded to treatment compared to 35.3% in the placebo group. This is a significant difference of 19.4% and compares well with other recent drugs approved for IBS-D – Viberzi (eluxadoline, Allergan (NYSE:AGN)) and Xifaxan (rifaximin, Valeant), for which the respective percentage rates were 13.5% and 10.5% (noting the limitations of making comparisons across different trials). Both Viberzi and Xifaxan were approved for IBS-D in 2015 and in 2016 had solid sales of $93m and $289m, respectively, in IBS-D (EvaluatePharma).
To read the entire report Please click on the pdf File Below: