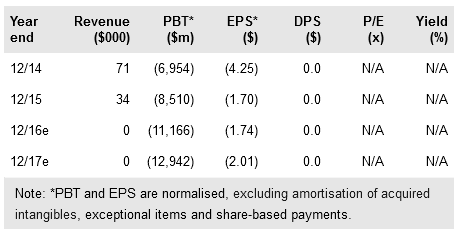
RXi Pharmaceuticals (NASDAQ:RXII) continues to make solid progress with its development pipeline. We anticipate one of two promising cosmeceutical candidates to enter human testing this year, moving forward on an expedited pathway for potential launch in 2019. We also expect data in H216 in all lead projects including RXI-109 in dermal scarring and in retinal scarring in wet AMD, and Samcyprone in cutaneous warts.
Cosmeceuticals move forward in skin appearance
In the latter part of 2015, RXi selected two treatment candidates to move forward on an expedited cosmeceuticals development pathway, RXI-231 (targeting tyrosinase to improve skin appearance) and RXI-185 (targeting collagenase for skin elasticity and anti-ageing). A successful cosmeceuticals launch could fetch sales of up to $150m and we expect at least one candidate to move into consumer testing by year end, for a targeted launch in 2019. RXi management has considerable expertise in cosmetic product development and we anticipate the completion of a series of marketing deals with large consumer product companies in coming years to bring in funds that can provide funding of RXi’s therapeutic pipeline.
To read the entire report Please click on the pdf File Below