Following-on from ReNeuron Group PLC (LON:RQE)’s recent striking early-stage data release for its human retinal progenitor cell (hRPC) product in retinitis pigmentosa (RP), and the ongoing partnering interest in cellular therapies, ReNeuron has partnered its two lead products – hRPC and the CTX cell line – for the Chinese market with Fosun Pharma. We have updated our model to reflect the near-term part of the £80m in potential total upfront payments and milestones.
Open season on cellular therapies
After the acquisition of two US chimeric antigen receptor T-cell (CAR-T) companies, Kite and Juno, in 2017 for $12bn and $8bn, respectively, cellular therapies have become a rich source of licensing and acquisition transactions. With two products in two clinical studies and an exosome platform, it was probably only a matter of time before ReNeuron announced its first deal of 2019. While ReNeuron’s first transaction covers two products for the Chinese market, the rest of the world remains available and it is unlikely that this will be the last such transaction it announces. Our model anticipates a further £10m transaction before year end 2020.
Fosun licenses ReNeuron’s lead products for China
In a well-received press release, ReNeuron announced the licence of its two lead products – the CTX and hRPC cell therapy programmes for chronic stroke and degenerative retinal diseases, respectively – to Shanghai and Hong Kong-listed Shanghai Fosun Pharmaceutical Industrial for the Chinese market. The transaction includes a £6m upfront payment, an additional £6m in near-term operational milestones and a further £8m in future regulatory milestone payments that together with post-launch profit milestones, could be worth up to £80m in total. The deal also includes tiered royalties of between 12% and 14% of Chinese sales and Fosun is fully funding the development and commercialisation of ReNeuron’s products in China.
Valuation: Updated for the Fosun transaction
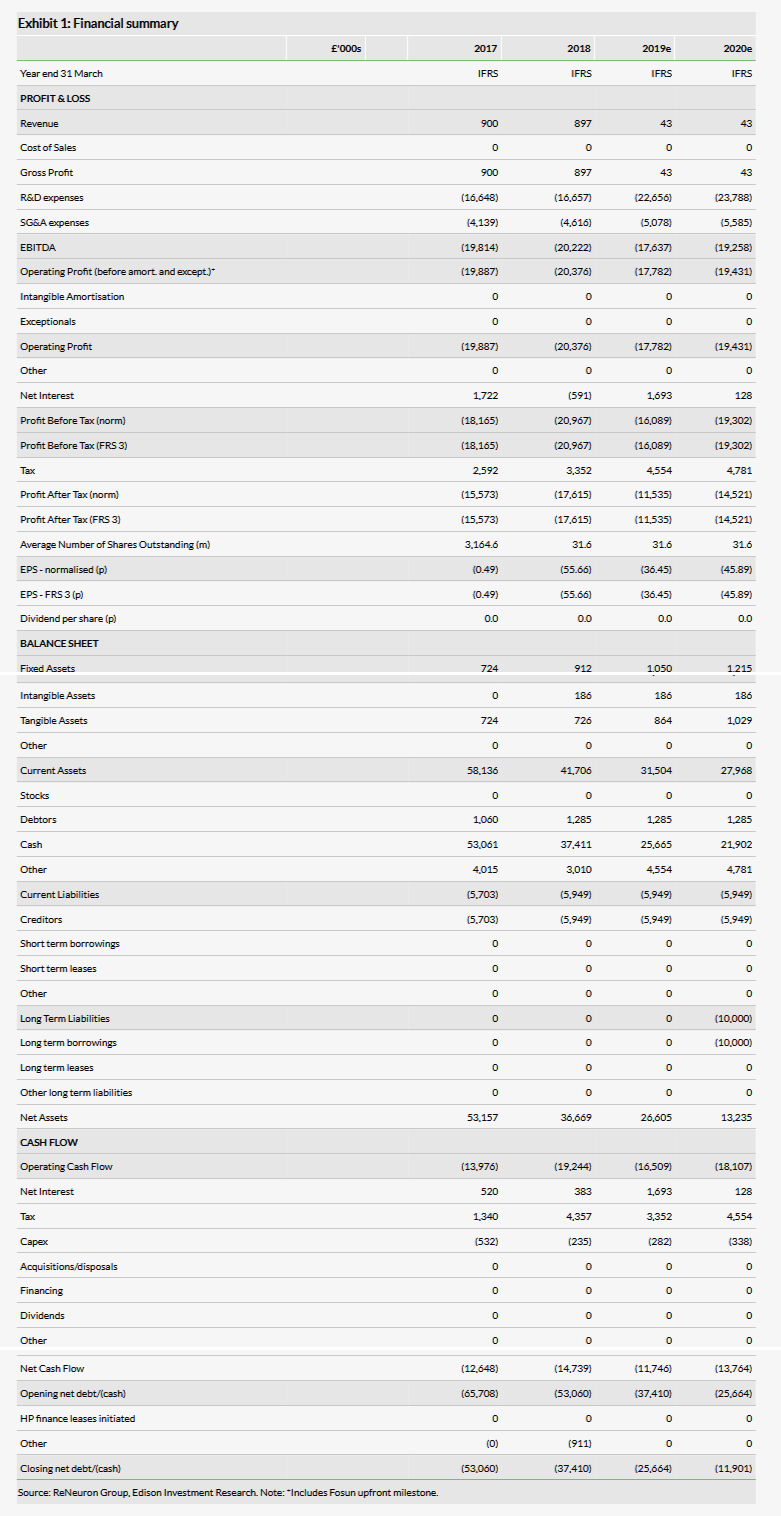
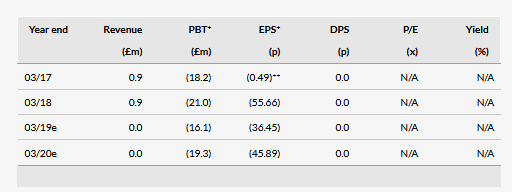
Our previous model did not include Chinese sales for ReNeuron’s CTX or hRPC products, so we have added these with some conservative assumptions for penetration and pricing in China, and have used the epidemiology from Japan. In our model we have approximated the announced milestone flow up to launch in China as other income, but left the probabilities of success for both CTX and hRPC unchanged at 25% and 20%, respectively. Our valuation moves from £192m or 608p per share, to £199m or 630p per share.
Business description
ReNeuron is a UK biotech company developing allogeneic cell therapies. The first pivotal Phase IIb trial for CTX neural stem cells for chronic stroke disability is underway. Human retinal progenitor cells (hRPCs) are also being studied for RP (in Phase I/IIa).