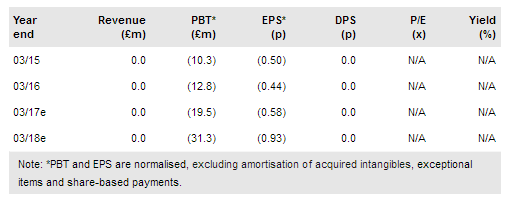
Reneuron Group (LON:RQE) continues to prepare for its Phase III chronic stroke study (due to start in H217) and has indicated that it is making progress with the US/EU regulatory authorities. It intends to submit an IND in the US in Q217 and shortly after in the EU. ReNeuron also announced that it now plans to expand its ongoing Phase I/II retinitis pigmentosa (hRP cells) study to 20 patients in Phase II and commence a Phase II study in a new indication: cone-rod dystrophy (CRD). As a result, it will not continue development of CTX in critical limb ischaemia (CLI). We maintain our rNPV at £291m.
Phase III chronic stroke study set-up progressing
ReNeuron reported positive Phase II trial data (December 2016) for its CTX cells in chronic stroke patients. It has recently announced that it is on track to initiate the follow-on Phase III trial in H217. It is currently consulting with the regulatory bodies in Europe and the US, has an end-of-Phase II meeting with the FDA shortly and intends to submit an IND in Q217. The European clinical trial application will follow shortly after. Alongside this, discussions are ongoing with the Japanese regulatory agency, which could offer a conditional marketing approval. For more detail, see our March 2017 outlook note.
To read the entire report Please click on the pdf File Below