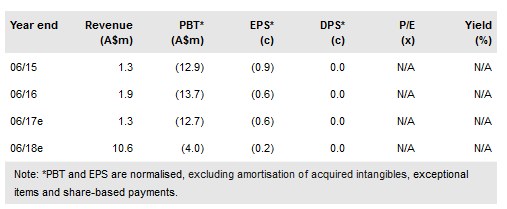
Prima BioMed (NASDAQ:PBMD) maintains its leadership in LAG-3 immunotherapies with the initiation of the randomised Phase IIb component of the AIPAC trial of IMP321 in breast cancer. In addition, the TACTI-Mel study of IMP321 combined with Keytruda in melanoma is expected to complete recruitment and report safety data in mid-2017, while the pipeline now includes a first-in-class Lag-3 agonist antibody. Initial efficacy data on the first 15 patients in AIPAC is also expected mid-year. We have revised development timelines for in-house and partnered LAG-3 programmes, which sees our valuation decrease to A$252m or A$0.12/share (vs A$282m or A$0.14/share).
Randomised AIPAC Phase IIb underway
In January, Prima dosed the first of 226 patients in the randomised Phase IIb component of the AIPAC trial in metastatic breast cancer (mBC). Patients will receive either 30mg of the IMP321 APC activator or placebo, in combination with paclitaxel. The 30mg dose is 5x higher than the dose that delivered a 50% response rate in an earlier study, and was well tolerated in the safety run-in study.
To read the entire report please click on the pdf file below