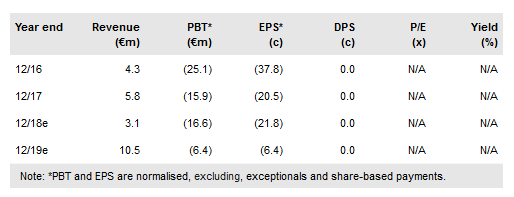
Paion O.N (DE:PA8G) and partner Cosmos Pharmaceutical took an important step towards a US filing for its ultra-short-acting sedative/anesthetic remimazolam, with the completion of a successful pre-NDA meeting in July. Filing in Japan (via partner Mundipharma) is expected before the end of 2018. Paion took a step towards its goal of self-commercializing remimazolam in Europe with the initiation of a Phase III study in general anesthesia (GA) in July. The company is funded beyond top-line data from the EU Phase III trial expected in 2019. We lift our valuation to €283m or €4.44 per share.
Successful pre-NDA meeting: US filing Q418 or Q119
Partner Cosmo has completed a successful pre-NDA meeting with the US FDA and remains on track to file for approval of remimazolam for procedural sedation in Q418 or Q119. Paion would receive a €7.5m milestone upon filing in the US.
To read the entire report Please click on the pdf File Below: