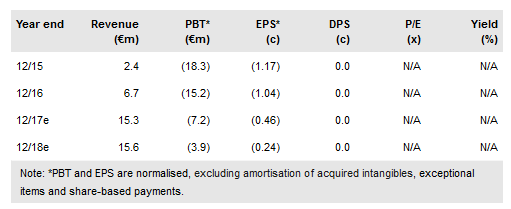
We anticipate that the sales and pipeline progression made by Newron Pharmaceuticals SpA (SIX:NWRN) in 2016 will be cemented further in 2017/18. A critical catalyst remains the US Xadago (Parkinson’s disease (PD) therapy) approval which is imminent (revised PDUFA date is 21 March); this will materially impact the share price performance. We anticipate US launch in H217 by sub licensee, US WorldMeds. Data from pipeline assets sarizotan (Rett’s syndrome, RS) and evenamide (schizophrenia) in the upcoming 18 months will highlight Newron’s diverse, innovative CNS-based R&D portfolio. We value the company at CHF530m.
Xadago sales building; US PDUFA date, 21 March
Newron reported €1.7m in Xadago royalties from commercial partner, Zambon in FY16 and Xadago is now available in 11 European countries. We anticipate US FDA approval on 21 March to trigger a €9m milestone payment to Newron. Zambon has sub-licensed US commercialisation rights to US WorldMeds and we anticipate US launch in H217. We therefore expect FY17 sales to benefit from the ongoing European market rollout by Zambon and the US launch by US WorldMeds.
To read the entire report Please click on the pdf File Below