Merck (NYSE:MRK) announced disappointing results from a pivotal phase III KEYNOTE-061 study, evaluating its anti-PD-1 therapy, Keytruda (pembrolizumab), as a second-line treatment for patients with advanced gastric or gastroesophageal junction (“GEJ”) adenocarcinoma.
Notably, Keytruda is already approved in the United States as a monotherapy for third-line treatment of advanced gastric or GEJ. Additionally, it is approved for many types of cancers and treatment settings including lung cancer, melanoma, head and neck cancer, classical hodgkin lymphoma and bladder cancer.
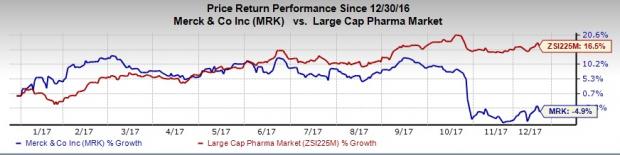
Shares of Merck has lost 4.9% this year so far compared unfavorably with the industry’s 16.5% increase.
The phase III study failed to meet its primary endpoint of overall survival (OS). Notably, the progression free survival (PFS) showed no statistical significance in the PD-L1 positive patient population.
However, the company said that it will continue to evaluate Keytruda in two other phase III studies, both as a monotherapy as well as in combination with chemotherapy as a first-line treatment for patients with GEJ.
We remind investors that Keytruda is a key top-line driver for Merck and it brought in $1.5 billion sales in third-quarter 2017, up 18.8% sequentially and 194% year over year. Sales continue to be driven by a launch of new indications globally.
Significantly, Keytruda sales in the United States have gained particularly from a strong momentum in the new indication of first-line lung cancer. The drug’s sales were primarily driven by melanoma outside the United States.
Meanwhile, the Keytruda development program also significantly advanced this year with regulatory approvals for four new indications in the United States and two additional diseases in Europe.
Important approvals include nods for advanced bladder cancer in both the United States and the EU, advanced microsatellite instability-high cancers and the first sanction for a combination therapy with Eli Lilly & Company’s (NYSE:LLY) cancer drug Alimta (pemetrexed) and carboplatin (pem/carbo), a commonly used chemo regimen in lung cancer.
Keytruda is continuously growing and expanding into new indications and markets globally. It is under trial for more than 30 types of cancer in above 650 studies including excess of 300 combination studies.
Merck is collaborating separately with several companies namely Amgen, Inc. (NASDAQ:AMGN) , Incyte, Glaxo and Pfizer, Inc. (NYSE:PFE) for the evaluation of Keytruda in combination with other regimens.
Zacks Rank
Merck carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Investor Alert: Breakthroughs Pending
A medical advance is now at the flashpoint between theory and realization. Billions of dollars in research have poured into it. Companies are already generating substantial revenue, and even more wondrous products are in the pipeline.
Cures for a variety of deadly diseases are in sight, and so are big potential profits for early investors. Zacks names 5 stocks to buy now.
Pfizer, Inc. (PFE): Free Stock Analysis Report
Eli Lilly and Company (LLY): Free Stock Analysis Report
Merck & Company, Inc. (MRK): Free Stock Analysis Report
Amgen Inc. (AMGN): Free Stock Analysis Report
Original post
Zacks Investment Research