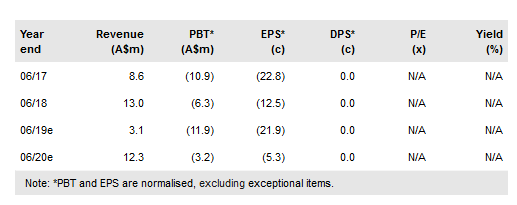
Kazia Therapeutics Ltd (AX:KZA) has added two further indications to the development programme for its brain-penetrant PI3K inhibitor GDC-0084 through collaborations with prestigious US-based cancer centres. The collaborations further validate the potential of GDC-0084, which was in-licensed from Genentech in 2016. Importantly, the two additional indications will provide alternative pathways to a potential first marketing approval for GDC-0084, increasing the overall likelihood of success. Kazia’s ongoing Phase IIa study of GDC-0084 in glioblastoma (GBM) is expected to report first data in early 2019. Kazia raised A$3.4m through a recent share placement and has a share purchase plan (SPP) underway to raise additional funds. We increase our valuation range to between A$83m and A$139m.
Breast cancer brain metastases with Dana-Farber
The first collaboration is with the Dana-Farber Cancer Institute to investigate GDC-0084 in combination with Herceptin in women with HER2-positive breast cancer who have developed brain metastases. We had already included this potential indication in our valuation model for GDC-0084, so we are pleased to see that a trial will soon be underway. Genentech showed that GDC-0084 improves survival in this indication in animal studies, and a successful Phase III study for Novartis’s BYL719 validates targeting PI3K in breast cancer.
To read the entire report Please click on the pdf File Below..