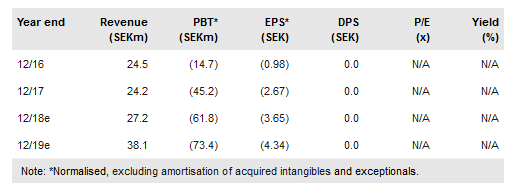
Immunovia publ AB (ST:IMMNOV) is preparing to market its IMMray PanCan-d for self-pay patients at high risk of pancreatic cancer (PC) in Q418, after preparatory activities were pushed back slightly from mid-2017 to 2018. To achieve reimbursement, a prospective trial in this population is being run and if positive, the test could be reimbursed in 2020. Immunovia is pursuing other groups ie patients with new-onset T2 diabetes aged 50+ and those with early gastric symptoms. Our revised valuation is SEK3.6bn as we now include early gastric symptoms. Autoimmune diseases represent upside.
First self-pay sales in sight in high-risk patients
Immunovia plans to market its lead indication the IMMray PanCan-d test in late 2018 in the private self-pay market following a slight delay to its pre-marketing activities from mid-2017 to this year. The company is also running the PANFAM-1 study, a three-year prospective clinical trial in 1,000 high-risk patients to achieve reimbursement. At SEK5,000/test and two tests per year, we estimate the total market opportunity is c SEK2bn in the EU/US, based on c 200k potential patients.
To read the entire report Please click on the pdf File Below: