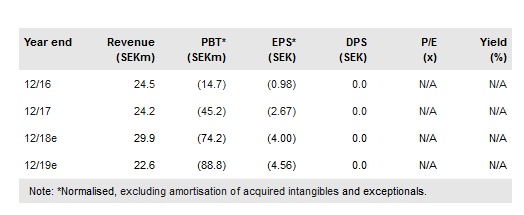
Immunovia publ AB (ST:IMMNOV) has announced that it needs to optimise the algorithms used in the IMMray PanCan-d test in high-risk patients due to sample collection-related variability, which will delay the first sales of the product for a year from late 2018 to late 2019. Separately, Immunovia has shown the IMMray platform can differentiate between healthy and lung cancer samples with 95% accuracy. On the autoimmune front, IMMray has proven efficacious in detecting patients with rheumatoid arthritis (RA) who tested negative for antibodies against cyclic citrullinated peptides (CCP) with 90% accuracy, allowing a potential increase of up to 30% in detection rate. Both studies will be validated in further trials. Net cash at end H118 was SEK447.2m. Our updated valuation is SEK3.5bn vs SEK3.6bn before.
PanCan-d first self-pay sales delayed for a year
In retrospective analysis of 1,000 samples, Immunovia found that different blood-collection procedures introduced variability in the test algorithms. Therefore, additional optimisation work is needed, which the company estimates will cost SEK5m. The interim analysis of the prospective PANFAM-1 clinical trial in high-risk individuals will now be conducted in Q119 (vs Q318 before); however, the company still expects to complete the interventional phase during 2019. We continue to estimate the total market opportunity is SEK2bn in the EU/US, based on 200,000 potential patients.
To read the entire report Please click on the pdf File Below: