BioLineRx Ltd. (NASDAQ:BLRX) announced that it has completed patient recruitment in the triple combination arm of the ongoing phase IIa COMBAT/KEYNOTE-202 study on Motixafortide (BL-8040) for treating second-line metastatic pancreatic cancer. The candidate, a CXCR4 inhibitor, is currently being evaluated in combination with Merck’s (NYSE:MRK) PD-L1 inhibitor Keytruda (pembrolizumab) and chemotherapy for addressing the given indication.
Per the press release, the primary endpoint of the study is to see the objective response rate (ORR) while the secondary endpoints include overall survival, progression free survival and disease control rate (DCR). Patients were treated with Motixafortide monotherapy priming treatment for five days followed by combination cycles of chemotherapy (Onivyde/5-fluorouracil/leucovorin), Keytruda and Motixafortide until progression.
BioLineRx plans to announce progression-free and overall survival data from the triple combination arm of the above-mentioned study in mid-2020.
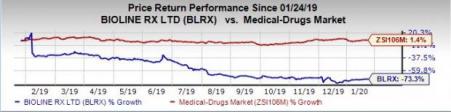
Shares of BioLineRx have plunged 73.3% in the past year against the industry’s increase of 1.4%.
We remind investors that in December 2019, BioLineRx reported preliminary data from the triple combination arm of the phase IIa COMBAT/KEYNOTE-202 study, which showed that, treatment with the triple combo led to a 32% ORR and 77% DCR from the 22 patients evaluated at the time.
Earlier this month, Motixafortide was granted an orphan drug designation by the European Commission (EC) for the treatment of pancreatic cancer. The candidate already received the same designation in the United States from the FDA for treating the same indication.
Notably, BioLineRx has a collaboration agreement with Genentech, a unit of Roche (OTC:RHHBY) , for evaluating Motixafortide in combination with Tecentriq (atezolizumab) in two phase Ib/II studies for solid tumor.
Zacks Rank & Key Pick
BioLineRx currently carries a Zacks Rank #3 (Hold). A better-ranked stock in the same sector is KalVista Pharmaceuticals, Inc. (NASDAQ:KALV) , sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
KalVista’s loss per share estimates have narrowed 17.6% for 2020 over the past 60 days. The stock has gained 6.8% in the past year.
The Hottest Tech Mega-Trend of All
Last year, it generated $24 billion in global revenues. By 2020, it's predicted to blast through the roof to $77.6 billion. Famed investor Mark Cuban says it will produce "the world's first trillionaires," but that should still leave plenty of money for regular investors who make the right trades early.
See Zacks' 3 Best Stocks to Play This Trend >>
Merck & Co., Inc. (MRK): Free Stock Analysis Report
Roche Holding (SIX:ROG) AG (RHHBY): Free Stock Analysis Report
BioLineRx Ltd. (BLRX): Free Stock Analysis Report
KalVista Pharmaceuticals, Inc. (KALV): Free Stock Analysis Report
Original post
Zacks Investment Research
