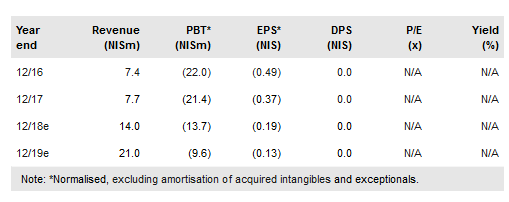
Allium Medical Solutions Ltd (TA:ALMD) has announced FDA approval of its subsidiary Gardia’s Wirion, which has become the only embolic protection system approved for all atherectomy procedures. Allium has reported FY17 results, with revenues up 5% y-o-y to NIS7.7m, and announced its first order in Mexico for NIS300k and approval of some of its stents in Russia. However, registration of the remaining stents and IBI Medical (EndoFast soft tissue fixation) in Mexico and Russia and stents in China has been delayed; we believe approval is possible in 2018. Mexico, Russia and China are the bulk of the distribution deals (NIS132m) and delays are the major reason for the slight decline in our valuation to NIS1.64/share (NIS1.68/share previously).
FDA approves Wirion; chances of transaction higher
Gardia Medical’s Wirion system has received FDA approval for leg artery catheterisation on the back of strong clinical data from the WISE-LE trial, which met its primary and secondary endpoints at interim analysis. Wirion captured a significant amount of debris in all clinical cases and across all atherectomy devices and is now the only embolic protection system cleared by the FDA for all atherectomy procedures. Allium is looking for a strategic transaction, preferably M&A; we believe the FDA approval could make Gardia more attractive to potential partners. We model Wirion as part of Allium’s overall business and valuation and project revenue of NIS2.4m after full launch in 2018, rising to NIS8.6m in 2020 as we adjust our price forecast based on the most recent FY17 data.
To read the entire report Please click on the pdf File Below: