Addex Therapeutics Ltd. (ADDXF) is now focused on developing its clinical pipeline for rare diseases and will significantly reduce discovery activities. In 2013 Addex will complete a pilot Phase II study of dipraglurant for rare dystonia and target a partnership for the Parkinson’s disease (PD) indication. It will also complete a Phase I study of ADX71441 for Charcot-Marie-Tooth (CMT) disease, as well as select a clinical candidate for the mGlu4 PAM programme in multiple sclerosis. While Addex is currently financed to end-2013, potential licensing deals and/or financings could extend the cash runway.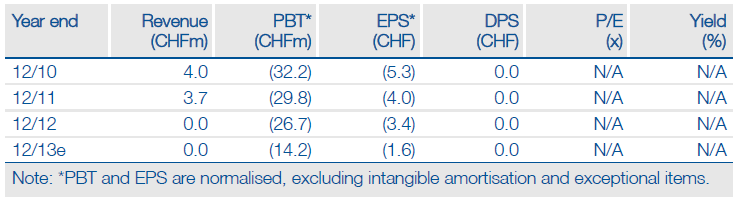
0BRefocusing, restructuring and realigning
Following a strategic review, Addex will now focus on developing its clinical-stage pipeline in rare (orphan) disease indications. The restructuring of research operations (suspension of discovery activities) will free up financial resources to implement this new strategy. Finally, Addex has also announced plans to list on a US stock exchange (ADR on NASDAQ), which could broaden the potential US investor base.
1BDipraglurant: Pilot Phase II in rare dystonias
The rationale for dipraglurant in dystonia is based on positive preclinical data and initial clinical findings in the Phase II study in PD, where the drug reduced levodopa-induced dystonia. We understand a Phase II trial in dystonia (start Q213) will readout in Q413. Addex will seek to partner the PD levodopa-induced dyskinesia (PD-LID) indication.
2BADX71441: Advancing into Phase I for CMT1A
ADX71441 is being developed for CMT Type 1A (CMT1A) disease, a rare inherited neurological disease. A Phase I study in healthy volunteers (start Q213) will assess the safety, tolerability and initial biological activity of ADX71441. Provided results are positive in Q413, the drug could enter a Phase IIa study in CMT1A patients in 2014.
3BCash runway to end-2013
Addex had cash of CHF15.3m at year-end 2012. We now model CH15m in operating expenditure for 2013, including restructuring related costs. On our revised projections, and barring new partnerships and/or financing, Addex is financed to year-end 2013.
4BValuation: Risk-adjusted NPV of CHF218m
We value Addex at CHF218m ($236m) or CHF25.40 per share. Our rNPV assumes industry-standard success rates for drugs based on their development stage and a hypothetical 18% royalty on dipraglurant and 12% on JNJ-40411813.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Addex: Refocusing, Restructuring And Realigning
Published 03/06/2013, 07:14 AM
Updated 07/09/2023, 06:31 AM
Addex: Refocusing, Restructuring And Realigning
New orphan disease focus
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.