Vernalis’s (VER.L) goal of achieving sustained profitability depends on the successful development of up to six prescription-only cough cold formulations for the US market. This portfolio is on track to deliver its first product for the 2015/16 winter season, with a further four formulations also progressing well. Tight cost control, coupled with a healthy cash balance of £85.7m, means Vernalis is funded through to profitability.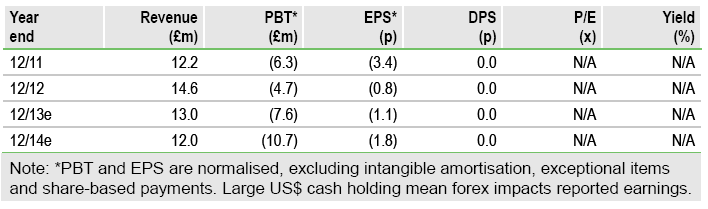
Broad cough cold portfolio building nicely
The Tris collaboration to develop up to six extended-release (ER) liquid formulations should be the formative first step that brings Vernalis to profitability. The US prescription-only (Rx) cough cold market provides a particularly attractive revenue opportunity, with high technological barriers to competitor entry. The first product, CCP-01, has successfully completed proof-of-concept and should be filed in mid-2014 with launch in 2015. Four other products are in active development.
Realising value from legacy programmes
Vernalis is also delivering on other aspects of its three-tier business strategy (commercial, development, research), with value extracted from both the existing NCE product pipeline and the abilities of the research team. The likely star of the development portfolio is AUY922, which is partnered with Novartis and is in 25 clinical studies in eight different cancer types. The research collaborations have also progressed well, with £2.5m in milestones received during H113.
Funded through to self-sustaining profitability
Vernalis is well funded, with a robust net cash position of £85.7m (June 2013). Continuing revenue streams (royalties from frovatriptan, milestone income and research collaboration funding), coupled with tight cost control, means Vernalis should have sufficient resources to build the necessary US cough cold marketing infrastructure and fund the remaining development activities through to profitability.
Valuation: Ex-cash rNPV of £121m vs EV of c £42m
Our valuation for Vernalis is £206m with cash included (an ex-cash value of £121m). This consists of a conservative £98m rNPV of the US Rx ER cough cold product portfolio, £12m for the frovatriptan royalty NPV and an rNPV of £11m for the existing pipeline. Further upside could come from more clarity on cough cold portfolio progress, securing complementary products for the US, progress with the clinical pipeline and existing or new research collaborations.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Vernalis Company Proposition Gathering Momentum
Published 09/24/2013, 08:06 AM
Updated 07/09/2023, 06:31 AM
Vernalis Company Proposition Gathering Momentum
Value proposition gathering momentum
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.