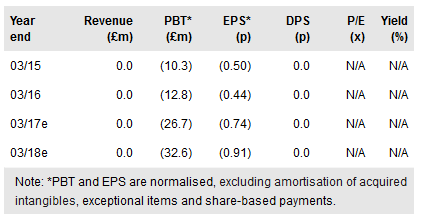
Investor focus is currently on the readout of ReNeuron’s Phase II stroke disability study with its CTX cells, due in Q416. Alongside this there are a number of other data readouts and progression of its pipeline potentially in the near term. Reneuron Group (LON:RQE) has significant financial resources to support its broad clinical development programme over the next two to three years. Adjusting for cash, strengthening of the $:£ exchange rate and revised clinical timeline for CTX in stroke (patient recruitment completed in June 2016), we raise our rNPV to £249m (vs £233m).
Well-funded for pipeline development
ReNeuron has the funds to continue to significantly advance its cell-based therapies: CTX, hRPC and exosomes. CTX stem cells are currently in a Phase II study for stroke disability and a Phase I safety trial for critical limb ischaemia (CLI). The hRPC (human retinal progenitor cells) candidate is in a US Phase I/II study for retinitis pigmentosa (RP). Finally, ReNeuron has recently selected the early-stage exosome programme’s initial clinical target, glioblastoma multiforme (GBM).
To read the entire report Please click on the pdf File Below