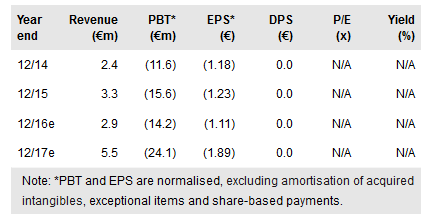
Pixium Vision (PA:PIX) announced on 25 July that its Iris II epi-retinal implant has received CE mark clearance from European regulatory authorities. This pivotal step helps validate the device’s safety and can enable the first stage of a commercial roll-out of this device in certain European markets. Our rNPV, inclusive of net cash, is €11.62 per share (vs €10.28 previously).
Clinical trial and reimbursement activities underway
We expect Pixium to implement the first phase of an EU commercial roll-out and seek reimbursement in Germany, France, the UK, Spain and Austria. Emphasis will be placed on selected ophthalmic surgery centres of excellence, particularly those sites that have participated in earlier trials of the earlier-generation Iris I implant.
We believe that Second Sight’s Argus II implant is reimbursed at €96,000 in France (Pixium’s home market). We believe this can bode well for Pixium, as the Iris II has significantly more electrodes than the Argus II (150 vs 60), which may improve the level of visual resolution perceived by the user.
To read the entire report Please click on the pdf File Below