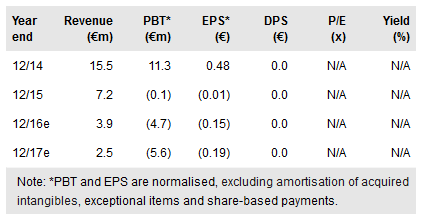
With its Q216 results, Oryzon Genomics SA (MC:ORY) reported that the two ongoing clinical studies, involving clinical-stage products ORY-1001 and ORY-2001, are proceeding as planned. Preliminary data from the Phase I/IIa trial with ORY-1001 are due by end-2016, which is the main catalyst for the company in the near term. ORY-2001 demonstrated safety when administered in a single dose and is now being tested in multiple ascending doses in Phase I. Recently Oryzon expanded its R&D pipeline with a new preclinical asset leveraging its proprietary epigenetics platform. Our valuation is virtually unchanged at €158m or €5.5/share.
Q216 results in line, solid cash position
Oryzon reported its Q216 results on 8 August. R&D expenses were €2.2m, slightly up year-on-year. G&A costs were €2.1m (Q215: €1.6m) due to new personnel hires and increased other expenses after the IPO in December 2015. Solid cash and term deposits of €30.1m (net cash of €7.1m) at the end of Q216 were significantly boosted with the new debt of €10.5m in May. Oryzon has a history of efficient use of available public grants, which could provide further non-dilutive financing.
To read the entire report Please click on the pdf File Below