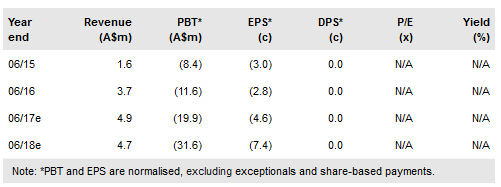
Novogen Ltd (AX:NRT) has in-licensed global rights to the Phase II-ready drug GDC-0084 from Genentech. GDC-0084 is designed to treat brain cancers, including the rapidly fatal glioblastoma (GBM). Novogen plans to initiate a Phase II trial in GBM next year and positive trial results could allow filing for accelerated approval in 2020. The transaction will complement Novogen’s preclinical assets and Cantrixil, which will enter Phase I in ovarian cancer in Q416. Our valuation of Novogen’s existing programme is unchanged at A$112m (A$0.26/share). We value GDC-0084 under two different scenarios at A$11-54m, after accounting for A$35m of anticipated clinical trial costs.
GDC-0084: PI3K inhibitor targeting brain cancer
GDC-0084 is a small molecule phosphoinositide 3-kinase (PI3K) inhibitor, which readily crosses the blood-brain barrier and exhibited dose-dependent reductions in tumour growth in Phase I. The Phase II trial is likely to test GDC-0084 as first-line maintenance chemotherapy in newly diagnosed GBM following surgical resection and six weeks of chemoradiotherapy. The trial will recruit a biomarker-defined subgroup of patients who receive little benefit from standard chemotherapy.
To read the entire report Please click on the pdf File Below