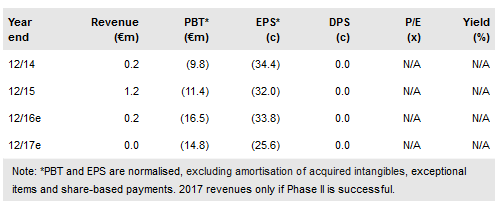
Neovacs (PA:ALNEV) is running a Phase IIb trial on its lead immunotherapy project, IFN-Kinoid (IFN-K) for lupus. The US arm is now recruiting and is expanding from five to 15 centres due to strong interest. We now expect results from end 2017, formerly mid-2017. In Korea, CKD may start sales in 2018, but the main impact is likely to be in 2019. Partnering for other territories is now expected from mid-2018. The important VEGF-Kinoid for cancer and AMD might start Phase I in H117. Cash at the end of June 2016 was €9.2m.
Ph IIb data from late 2017; partnering from mid-2018
The US arm of the 178-patient study is being expanded from five to 15 centres due to strong clinical interest. Neovacs indicates that it expects to complete overall recruitment in Q117. With a 34-week (nine-month) follow-up, we now expect trial data in late 2017 or Q118, formerly mid-2017. We still expect CKD to launch in Korea in 2018, but the main launch will be in 2019. Neovacs intends to partner IFN-K, probably from mid-2018. We estimate that this could enable regulatory filings by 2021 and possible US and EU marketing from 2022, formerly 2021.The trial assesses the interferon signature, seen as a key lupus marker, and measures BILAG clinical response scores.
To read the entire report Please click on the pdf File Below