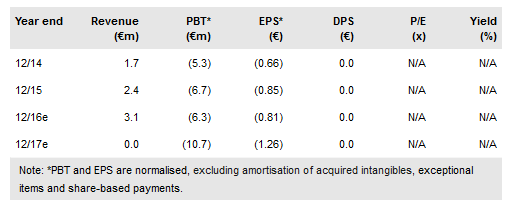
As part of its revised strategic plan, Crossject (PA:ALCJ) has expanded its partnership with CENEXI to fill and finish all of Crossject’s products in all regions for a period of five years, with a potential extension up to 10 years. Also, the timelines for its expected regulatory submissions have shifted across the board; most notably, the EU filing for ZENEO Methotrexate has been pushed back from H216 to H218 due to its non-emergency nature and higher requirement for manufacturing resources than other products. As a result, we reduce our valuation to €9.91 per share (from €12.07).
CENEXI deal expanded
Originally, the CENEXI agreement was for three years and focused on ZENEO Methotrexate for the Indian and Chinese markets. It has now expanded to cover all products in all regions for a period of five years with a potential extension up to 10 years. Manufacturing of clinical batches should begin in Q217, with a mass production line capable of filling up to 10m units (a number greater than our peak estimates) available in H118.
To read the entire report Please click on the pdf File Below