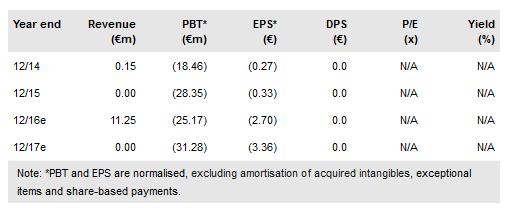
Celyad SA (NASDAQ:CYAD) has noted that the Phase I safety study on its NKR-2 CAR T-cell autologous therapy produced some “reports of clinical benefit”. The THINK Phase Ib trial has been approved in Belgium and awaits FDA clearance. This is a major expansion of CAR therapy with five solid tumours plus AML and MM being explored. As a result, we have raised the probability of success to 20% from 18.5%. There is a challenge to the granted 2009 US patent on allogeneic CAR T-cells. While the claim is being re-examined, the patent remains in force; other patents and patent applications provide protection. Edison’s interim indicative value has been rebased and increased to €45 per share, formerly €41 per share.
NKR-2 takes a lead positon in solid tumours
In the completed Phase I, Celyad’s CEO noted “reports of unexpected clinical benefit”: unexpected because the single, low doses were not expected to show efficacy; data will be presented at ASH in early December. Celyad now has Belgium approval to start immuno-oncology autologous NKR-2 trials (THINK) in two haematological and five solid tumours at high doses. The exploration of NKR-2 in solid tumours puts Celyad in a leading position in this area. Other CAR companies will have to compete for a limited number of patients in the congested CD19 area.
To read the entire report Please click on the pdf File Below