Vacc-4x continues to advance; next data in Q414
Bionor Pharma ASA (OSLO:BIONOR) Vacc-4x development remains on track, with ‘Kill & Boost’ data still expected in Q414 and data from the next part of the ongoing Reduc (‘Kill & Kick’) trial anticipated during 2015. Additional ‘Kick’ data from the Reduc study recently made a big splash at the AIDS 2014 conference. Cash should be sufficient to fund operations through completion of all ongoing clinical trials. Our valuation is increased slightly to NOK901m.
Reduc data well received at AIDS 2014 conference
Detailed data from both part A of the ongoing Reduc (‘Kick & Kill’) Phase I/II trial of Vacc-4x with romidepsin and from the previously reported Vacc-4x Reboost trial were recently presented at the International AIDS conference. Data from the Reduc trial are the first time that romidepsin has been shown to reactivate HIV viral reservoirs, which we believe is a critical step in the quest towards a functional cure. These data were well received at the conference, with Professor Steven Deeks, a key opinion leader in this field, commenting that this was “the single most important advance of this meeting and it will have a major impact in the future”. Activation of these undetectable viral reservoirs was triggered by romidepsin, with virus production in HIV-infected cells increasing 2.1 to 3.9 fold above normal and with measurable viral load in five of six patients. The ability to reawaken these reservoirs will be a key component of any cure strategy and during 2015 we expect data from part B of the Reduc trial, with reservoir size data in H115 and the effect on viral load
control during cART interruption in H215.
Revlimid combination data will provide insights
Bionor with partner Celgene is investigating ways to strengthen and boost the immune system to potentially amplify and maximise Vacc-4x’s immune response. This approach could form part of the cure strategy. The ‘Kill & Boost’ Phase II trial of Vacc-4x with the immune modulator (IMiD) lenalidomide (Revlimid) is ongoing and topline data from this small trial are anticipated during Q414. These data could provide insights into the use of IMiD combination therapy and any role that it might play towards a cure, helping to shape future development strategies.
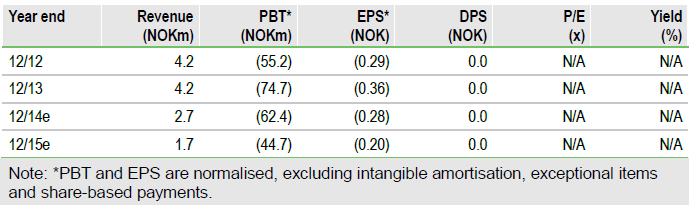
Valuation: Risk-adjusted NPV of NOK901m
Our underlying Vacc-4x assumptions are unchanged, with our rNPV now NOK901m (from NOK888m) owing to updated net cash and rolling our valuation forwards. We estimate net cash of NOK68m (at end June) should be sufficient to fund operations to end 2015, which should allow for completion of all ongoing trials. Data from these will be key to shaping Vacc-4x’s future development path.
To Read the Entire Report Please Click on the pdf File Below