Baseline patient characteristics and the ZFP64 biomarker correlate with improved survival in liver cancer (HCC) subjects receiving resminostat. These findings will guide 4SC’s, (FSCGF) pivotal trial plans in front-line HCC. We are optimistic that 4SC can secure financing/partner to conduct the Phase IIb/III trial and model €789m peak sales. Our rNPV is unchanged at €122m.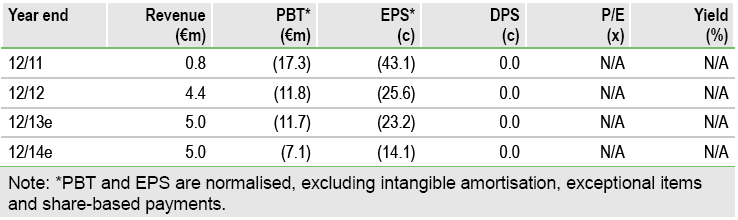
ECCO presentation of resminostat data
We attended the European Cancer Congress (ECCO) where 4SC presented a subgroup analysis from the SHELTER Phase II trial in advanced hepatocellular carcinoma (HCC). The post hoc analysis showed that baseline patient factors and a blood-based biomarker, ZFP64 (zinc-finger protein 64), correlate with improved overall survival (OS) following resminostat. 4SC will incorporate these findings into its pivotal Phase IIb/III trial plans for the resminostat/sorafenib combination in HCC.
Baseline patient factors predict OS benefit
Baseline characteristics correlating with improved OS include good performance status (ECOG 0), absence of liver cirrhosis (Child-Pugh A) and lack of vascular invasion. We expect 4SC’s planned front-line Phase IIb/III trial in advanced HCC patients (BCLC stage C) to incorporate these findings as enrolment (Child-Pugh A) or stratification (ECOG, vascular invasion) criteria. Importantly, these criteria (BCLC-C, Child-Pugh A) mirror the clinical use of sorafenib in front-line HCC.
Increasing evidence for ZFP64 biomarker
High baseline ZFP64 levels (seen in c 60% of HCC patients) correlated with a significant (p=0.04) doubling of median OS vs low levels. Resminostat also down-regulated ZFP64 levels in peripheral blood of HCC patients, a finding confirmed by preclinical studies. This supports a link between HDAC activity and ZFP64 involvement in HCC cancer cell signalling. Although ZFP64 requires prospective validation, resminostat could become a targeted HCC therapy, with c 60% of patients stratified for treatment using a simple, blood-based companion diagnostic.
Valuation: rNPV remains at €122m
We value 4SC at €122m, or €2.41 per share, based on a risk-adjusted NPV analysis. Our rNPV includes resminostat at €108.5m, an indicative contribution from Phase I assets of €20m, year-end net cash of €4.5m, and deducts €11m of central costs. Potential Phase IIb development (vidofludimus) or divestment of non-core programmes (4SC-203, 4SC-207) would represent upside to our valuation.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
4SC: Baseline Patient Factors Predict OS Benefit
Published 10/13/2013, 04:53 AM
Updated 07/09/2023, 06:31 AM
4SC: Baseline Patient Factors Predict OS Benefit
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.