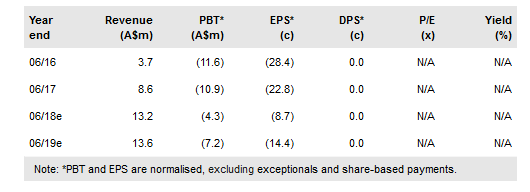
Kazia Therapeutics Ltd ADR (NASDAQ:KZIA) has initiated the dose optimisation lead-in component of the Phase II trial of GDC-0084 in glioblastoma, with initial data expected in Q119. Initial data from the Cantrixil ovarian cancer Phase I are due shortly. In January Kazia received a ~5% shareholding in Noxopharm (current market value ~A$4m) in return for collaborative support of that company’s lead programme. We roll forward our DCF model and add in the Noxopharm shareholding, which increases our valuation to between A$73m and A$133m.
GDC-0084 Phase II lead-in underway
Kazia has initiated a Phase II study of its oral PI3K inhibitor GDC-0084 in recently diagnosed glioblastoma (GBM). Genentech’s Phase I study of the drug was in sicker patients whose disease had relapsed after treatment, so Kazia’s trial includes a lead-in component to test whether newly diagnosed patients can tolerate higher doses of the drug, before the randomised study begins. The open-label lead-in should provide an initial data readout in early 2019.
To read the entire report Please click on the pdf File Below: